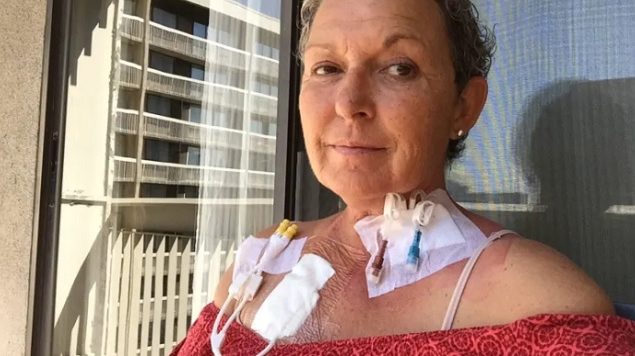
Canada’s health department has announced it intends to suspend its licences for Biocell breast implants following a safety review of breast implants and a rare form of cancer. Health Canada has been notified of 28 confirmed cases of BIA-ALCL, a serious form of non-Hodgkin lymphoma and of those, 24 involve Allergan’s Biocell implant.
The department is giving Allergan 15 days to present any new evidence about the safety of its textured breast implants. It says that “if a satisfactory response is not received by then,” then it will suspend the license and the product could no longer be sold in Canada.

Canada advises people wanting to have breast implants to talk to their doctor about the risks. (iStock)
Full review of breast implants coming
Health Canada says the entirety of its safety review of breast implants and this form of cancer will be available by the end of April.
It suggests that people with breast implants should consult a health care professional if they experience unusual changes to their breasts, including breast pain, sudden swelling, or a lump. It also says people wanting to get or remove implants should talk to their doctors.
More information is available on a government website about breast implants.
Canada has contacted France’s regulator about its decision today to suspend the sale of all macro-textured breast implant and polyurethane-coated breast implants. Allergan’s Biocell implants are the only macro-textured devices available in Canada. There are no polyurethane-coated breast implants available in Canada.






For reasons beyond our control, and for an undetermined period of time, our comment section is now closed. However, our social networks remain open to your contributions.