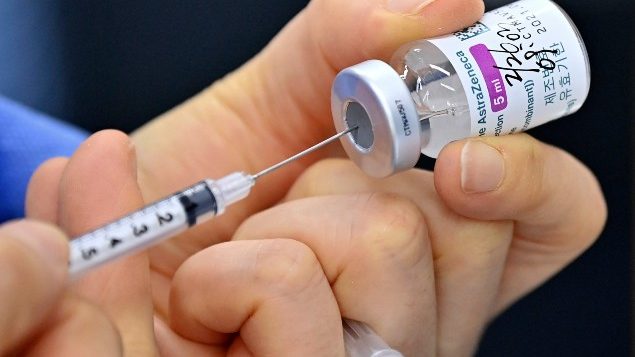
Canada’s health department has approved the AstraZeneca vaccine enabling health authorities to receive shipments within days and add it to vaccines already being administered from Pfizer and Moderna. Canada has pre-ordered 20 million doses and will get up to 1.9 million more AstraZeneca doses by the end of June 2021 through the global vaccine initiative called COVAX.
The AstraZeneca vaccine has an efficacy rate of 62 per cent which is lower than those of Pfizer and Moderna which are over 90 per cent. That means, someone getting the Pfizer or Moderna shot has an over 90 per cent chance of not getting COVID-19. While studies show that the AstraZeneca vaccine only provides a 62 per cent assurance of not contracting the disease, experiences of those who have already received it, show they are protected from severe illness, hospitalization and death. It is not clear yet what protection it provides against variants.
AstraZeneca vaccine easier to store and administer
Health authorities say some vaccination is better than none and that the AstraZeneca vaccine will be easier to roll out and administer because it does not require the very low storage temperatures that do the Pfizer and Moderna vaccines. This could especially benefit remote and northern communities in Canada.
There is also some question about how effective the AstraZeneca vaccines are for people 65 years old and older. Canada has a very large proportion of people in this age group. Because only 10 per cent of the people who took part in the company’s clinical studies, results are not conclusive. However, Canadian health officials again pointed to real world experiences, particularly in Scotland, that have shown the shots to be 95 per cent effective in preventing severe illness, hospitalization and death. They also point out that the goal for the efficacy of all vaccines is 50 per cent and the yearly influenza vaccination is only 64 or 54 per cent effective, yet it is recommended.

After vaccine deliveries ground to a near halt at the beginning of 2021, they finally picked up and people began lining up for a shot at the end of February. (Ryan Remiorz/The Canadian Press)
Advice pending for those 65 and older
Canadian regulators will also study the effects of mixing different vaccines, that is to say administering one type of vaccine as a first dose and a different one for the second. For now Health Canada is recommending both doses be from the same product.
A final recommendation on whether to use the AstraZeneca vaccine on people 65 years or older will soon come from Canada National Advisory Committee on Immunization. This committee is mandated to make “recommendations for the use of vaccines currently or newly approved for use in humans in Canada, including the identification of groups at risk for vaccine-preventable diseases for whom vaccination should be targeted.”
Because health care in Canada is delivered by the provincial and territorial governments, it is they who will ultimately look at their own demographics and decide who will get which vaccine.

The arrival of AstraZeneca vaccines may mitigate Prime Minister Justin Trudeau’s drop in popularity. (Justin Tang/The Canadian Press)
AstraZeneca approval may slow disapproval
News of the approval of the AstraZeneca vaccine comes as a great relief to Canadians. While the Canadian government was quick off the mark in pre-ordering vaccines from seven pharmaceutical companies, deliveries ground to a near halt in January and most of February 2021. This was a result of production delays as companies expanded their facilities to enlarge production capabilities. Canada relies on suppliers outside the country.
The delays moved Canada to a position of 40th among countries in terms of per capita vaccinations. Public opinion surveys suggest this prompted dissatisfaction among Canadians and lowered the popularity of Prime Minister Justin Trudeau. In the face of that, the prime minister promised the rate of vaccination would pick up dramatically and that all Canadians who want to be inoculated would be by the end of September 2021.
Vaccine roll-out may have election implications
Trudeau’s Liberal Party governs with a minority of seats in the House of Commons and rules only with the support of other parties. Minority governments usually last less than two years in Canada. Should the Trudeau government soon fall or wish to seek re-election, efficacy of the vaccine roll-out could play a major role in its fortunes.
Trudeau held a news conference to cheer the approval of the Astra-Zeneca vaccine. He announced that within weeks, two million of these doses will arrive and, by the end of March, vaccines from all three companies will number 6.5 million doses with tens of thousands more arriving between April and June.






For reasons beyond our control, and for an undetermined period of time, our comment section is now closed. However, our social networks remain open to your contributions.